NCERT Solutions for Class 10 Science Chapter 4 – Carbon and its Compounds
NCERT Solutions for Class 10 Science Chapter 4 – Carbon and its Compounds, contains solutions to various questions in Exercises for Chapter 4. Carbon and its Compounds Class 10 NCERT Solutions have been explained in a simple and easy-to-understand manner. NCERT Solutions for Class 10 help to check the concept you have learned from detailed classroom sessions and the application of your knowledge.
| Category | NCERT Solutions for Class 10 |
| Subject | Science |
| Chapter | Chapter 4 – Carbon and its Compounds |
Download NCERT Solutions for Class 10 Science Chapter 4 – Carbon and its Compounds
NCERT Solutions for Class 10 Science Chapter 4 – Intext Questions – Page 61
1. What would be the electron dot structure of carbon dioxide that has the formula CO2?
Answer:
Two oxygen atoms share their two outermost electrons with carbon to complete their octet.
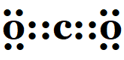
2.What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulfur? (Hint – The eight atoms of sulphur are joined together in the form of a ring.)
Answer:
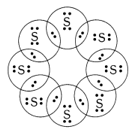
Intext Questions – Page 68
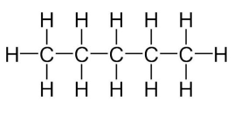
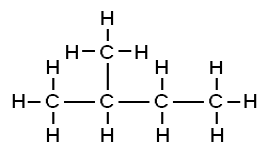
1. How many structural isomers can you draw for pentane?
Answer:
There are three structural isomers of pentane.
1. n-pentane
2. 2-methylbutane
3. 2,2 – dimethylpropane
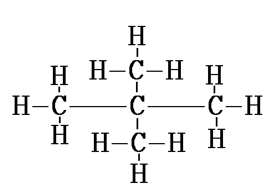
2. What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Answer:
The two properties of carbon which lead to the huge number of carbon compounds we see around us are:
(i) Carbon has high valency of 4. It requires 4 more electrons to complete its octet. So it can share electrons with a large number of other elements.
(ii) It is easy to from strong covalent bonds with carbon because of its small size.
3.What will be the formula and electron dot structure of cyclopentane?
Answer:
A cyclopentane has 5 carbon atoms and 10 hydrogen atoms. Each carbon atom shares 2 of its electrons with adjacent carbons and the rest two with 2 hydrogen atoms.
Formula: C5H10
Electron-dot structure:
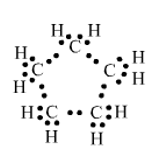
4. Draw the structures for the following compounds.
(i) Ethanoic acid
(ii) Bromopentane*
(iii) Butanone
(iv) Hexanal
*Are structural isomers possible for bromopentane?
Answer:
(i) Ethanoic Acid
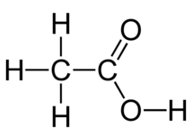
(ii) Bromopentane
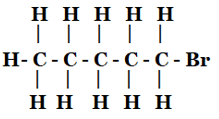
Yes, there are two other isomers of Bromopentane.
Names: 2-Bromopentane , 3-Bromopentane
(iii) Butanone
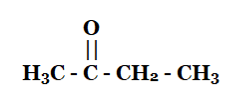
(iv) Hexanal
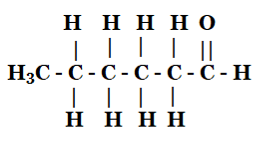
5. How would you name the following compounds?
(i) CH3—CH2—Br
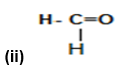
(iii )
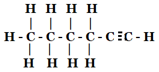
Answer:
(i) Bromoethane
(ii) Methanal or Formaldehyde
(iii) Hexyne
Intext Questions – Page 71
1. Why is the conversion of ethanol to ethanoic acid an oxidation reaction?
Answer:
Conversion of ethanol to ethanoic acid is an oxidation reaction because it involves the removal of hydrogen and the addition of oxygen.
Reactions Involved:
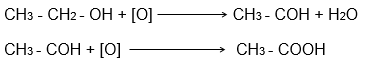
2. A mixture of oxygen and ethyne is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Answer:
When ethyne is burnt in oxygen, it burns completely and produces much more heat with a clean blue flame. If burnt in air, it will not combust completely leaving some residue and would not reach the required high temperature for welding.
Intext Questions – Page 74
1. How would you distinguish experimentally between an alcohol and a carboxylic acid?
Answer:
When we add sodium carbonate to carboxylic acid, we get a milky white color due to production of carbon dioxide gas. We don’t observe any such reaction in the case of alcohols.
The reaction involved is:
2CH3COOH + Na2CO3 → 2CH3COONa + H2O + CO2
2. What are oxidising agents?
Answer:
Agents that add oxygen and remove hydrogen from any compound are known as oxidising agents. Nitric acid, halogens, and potassium nitrate are examples of oxidising agents.
Intext Questions – Page 76
1. Would you be able to check if water is hard by using a detergent?
Answer:
No, we cannot check if water is hard by using a detergent. Detergents are salts of ammonium or sulphonates of long chain carboxylic acids. They can not react with magnesium and calcium present in hard water to form scum unlike soaps.
2.People use a variety of methods to wash clothes. Usually after adding the soap, they ‘beat’ the clothes on a stone, or beat it with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is agitation necessary to get clean clothes?
Answer:
People beat the clothes after adding soap to aid the micelles present in soap to trap and remove dirt, oil, and grease easily. On beating, the dirt particles dissociate the clothes and go away with water leaving the clothes clean.
NCERT Solutions for Class 10 Science Chapter 4 – NCERT Exercise – Page 77
1. Ethane, with the molecular formula C2H6 has
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds
Answer:
(b) 7 covalent bonds
2. Butanone is a four-carbon compound with the functional group
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) Alcohol
Answer:
(c ) Ketone
3. While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
(a) the food is not cooked completely.
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the fuel is burning completely.
Answer:
(b) the fuel is not burning completely
4. Explain the nature of the covalent bond using the bond formation in CH3Cl.
Answer:
Carbon can only form covalent bonds. It requires four more electrons to complete its octet. Three hydrogen atoms share their electrons and become stable and one chlorine atom shares its electrons to complete the octet of both carbon and chlorine.
5.Draw the electron dot structures for
(a) ethanoic acid
(b) H2 S
(c) propanone
(d) F2
Answer:
(a) ethanoic acid
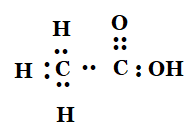
(b) H2S
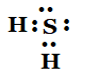
(c) propanone
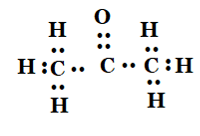
(d) F2
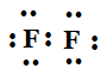
6. What is a homologous series? Explain with an example.
Answer:
Homologous series are a series of compounds which have the same functional group. They have similar general formula and chemical properties.
Physical properties of such compounds differ because of different molecular masses. For example, ethene, propene, etc. are all part of the alkene homologous series. Similarly, Methanol, ethanol, propanol, etc are the part of homologous series of alcohols.
7. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Answer:
Ethanol (C2H5OH) and ethanoic acid (CH3COOH) can be differentiated as follows:
| Ethanol | Ethanoic Acid |
|---|---|
| Does not react with sodium hydrogen carbonate. | Business Studies |
| Burning state. | Sour taste |
| No change on litmus paper | Turns Blue litmus paper to red. |
| Smells good. | Also known as acetic acid, smells like vinegar. |
8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Answer:
Soaps are sodium or potassium salts of long chain fatty acids. These salts have a polar and a non-polar end. The polar end of the salt is hydrophilic in nature and the non-polar end is hydrophobic. When added to water, these molecules orient themselves such that the hydrophilic ionic end stays towards the water and the hydrophobic end made of hydrocarbons protrudes out of water. In this arrangement, hydrophobic tails are in interior of the cluster and the ionic ends are on the surface of the cluster. Clusters of such molecules trap the dirt and result in micelle formation.
No, micelle is not formed in solvents like ethanol because these salts get easily dissolved in ethanol.
9. Why are carbon and its compounds used as fuels for most applications?
Answer:
Due to high calorific value, carbon compounds that contain carbon and hydrogen atoms in majority, give optimum ignition temperature and release high amount of energy. It is also easy to control their rate of combustion. Hence, carbon and its compounds are used as fuels for most applications.
10. Explain the formation of scum when hard water is treated with soap.
Answer:
Hard water contains calcium and magnesium ions. A soap contains salts of sodium and potassium. When we add soap to hard water, the soap molecules react with the ions present in water and give calcium and magnesium salts. These are sticky and insoluble, and are known as scum.
11. What change will you observe if you test soap with litmus paper (red and blue)?
Answer:
Soaps are alkaline in nature. Since they are salts of metals, they will be basic. If we dip a red litmus paper in a soap solution, it turns blue. If we dip a blue litmus paper, it doesn’t change its color.
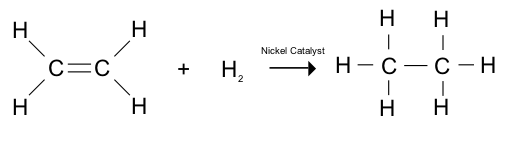
12. What is hydrogenation? What is its industrial application?
Answer:
Hydrogenation is the process by which hydrogen is added to a compound. It is a chemical reaction between hydrogen and another compound that needs to be reduced in the presence of a catalyst like platinum, palladium or nickel. Unsaturated hydrocarbons are converted into saturated hydrocarbons by this method. Its industrial application is to convert liquid vegetable oils and other compounds into solid or semi-solid fats.
13. Which of the following hydrocarbons undergo addition reactions: C2H6 , C3H8 , C3H6 , C2H2 and CH4 .
Answer:
Hydrocarbons that are not saturated and contain multiple bonds between two carbon atoms undergo addition reactions. Hence, C3H6 and C2H2 will undergo addition reaction.
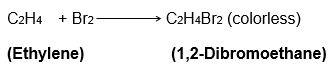
14. Give a test that can be used to differentiate between saturated and unsaturated hydrocarbons.
Answer:
Bromine water test can be used to differentiate between saturated and unsaturated hydrocarbons. It is an addition reaction and hence, only unsaturated compounds will undergo a change.
Bromine water is a reddish-brown colored solution made by dissolving bromine in water. When this solution is added to an unsaturated hydrocarbon ( such as alkenes and alkynes), its brown color disappears and the water is decolorised. No such change is observed when added to alkanes.
15. Explain the mechanism of the cleaning action of soaps.
Answer:
The soap molecules have different properties at different ends. One is hydrophilic end which dissolves in the water and is attracted towards the water and the second is the hydrophobic end which is dissolved in the hydrocarbons and is repulsive to water. The hydrophobic tail aligns itself along the surface of the water and is attracted to the hydrocarbons present in grease on dirty clothes. The cluster of such formation is called micelle. The oily dirt is collected in the middle of the micelle. The micelles stay in solution as colloid and form an emulsion. These are easy to wash away and leave clothes neat.
Topics Covered in Carbon and its Compounds Class 10 Science
- Bonding in Carbon-The Covalent Bond
- Versatile Nature of Carbon
- Saturated and Unsaturated Carbon Compounds
- Chains, Branches and Rings
- Homologous Series
- Nomenclature of Carbon Compounds
- Chemical Properties of Carbon Compounds
- Combustion
- Oxidation
- Addition Reaction
- Substitution Reaction
- Some Important Carbon Compounds-Ethanol and Ethanoic acid
- Soaps and Detergents
NCERT Solutions for Class 10 Science
- Chemical Reactions and Equations
- Acids, Bases and Salts
- Metals and Non metals
- Carbon and its Compounds
- Periodic Classification of Elements
- Life Processes
- Control and Coordination
- How Do Organisms Reproduce?
- Heredity and Evolution
- Light – Reflection and Refraction
- The Human Eye And Colorful World
- Electricity
- Magnetic Effects Of Electric Current
- Sources of Energy
- Our Environment
- Sustainable Management Of Natural Resources